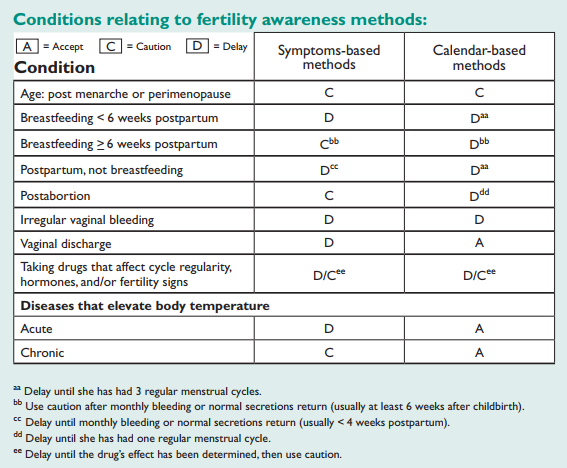
ADDITIONAL CONDITIONS
Additional conditions relating to emergency contraceptive pills:
Category 1: Repeated use; rape; CYP3A4 inducers (e.g., rifampicin, phenytoin, phenobarbital, carbamazepine, efavirenz, fosphenytoin, nevirapine, oxcarbazepine, primidone, rifabutin, St. John’s wort/Hypericum perforatum).
Category 2: History of severe cardiovascular complications (ischemic heart disease, cerebrovascular attack, or other thromboembolic conditions, and angina pectoralis)..
Additional conditions relating to female sterilization:
Caution: Diaphragmatic hernia; kidney disease; severe nutritional deficiencies; previous abdominal or pelvic surgery; concurrent with elective surgery
Delay: Abdominal skin infection; acute respiratory disease (bronchitis, pneumonia);systemic infection or gastroenteritis; emergency surgery (without previous counseling);surgery for an infectious condition; certain postpartum conditions (7 to 41 days after childbirth); severe pre-eclampsia/eclampsia; prolonged rupture of membranes (24 hours or more); fever during or immediately after delivery; sepsis after delivery; severe hemorrhage; severe trauma to the genital tract; cervical or vaginal tear at time of delivery); certain postabortion conditions (sepsis, fever, or severe hemorrhage; severe trauma to the genital tract; cervical or vaginal tear at time of abortion; acute hematometra); subacute bacterial endocarditis; unmanaged atrial fibrillation.
Special arrangements: Coagulation disorders; chronic asthma, bronchitis, emphysema, or lung infection; fixed uterus due to previous surgery or infection; abdominal wall or umbilical hernia; postpartum uterine rupture or perforation; postabortion uterine perforation.
Conditions relating to vasectomy:
No special considerations: High risk of HIV, asymptomatic or mild HIV clinical disease, sickle cell disease..
Caution: Young age; depressive disorders; diabetes; previous scrotal injury; large varicocele or hydrocele; cryptorchidism (may require referral); lupus with positive (or unknown) antiphospholipid antibodies; lupus and on immunosuppressive treatment.
Delay: Active STIs (excluding HIV and hepatitis); scrotal skin infection; balanitis; epididymitis or orchitis; systemic infection or gastroenteritis; filariasis; elephantiasis; intrascrotal mass.
Special arrangements: Severe or advanced HIV clinical disease may require delay; coagulation disorders; inguinal hernia; lupus with severe thrombocytopenia.
Conditions relating to male and female condoms, spermicides, diaphragms, cervical caps, and the lactational amenorrhea method:
All other conditions listed on the previous pages that do not appear here are a category 1 or NA for male and female condoms, spermicides, diaphragms, and cervical caps, and not listed in the Medical Eligibility Criteria for the lactational amenorrhea method.
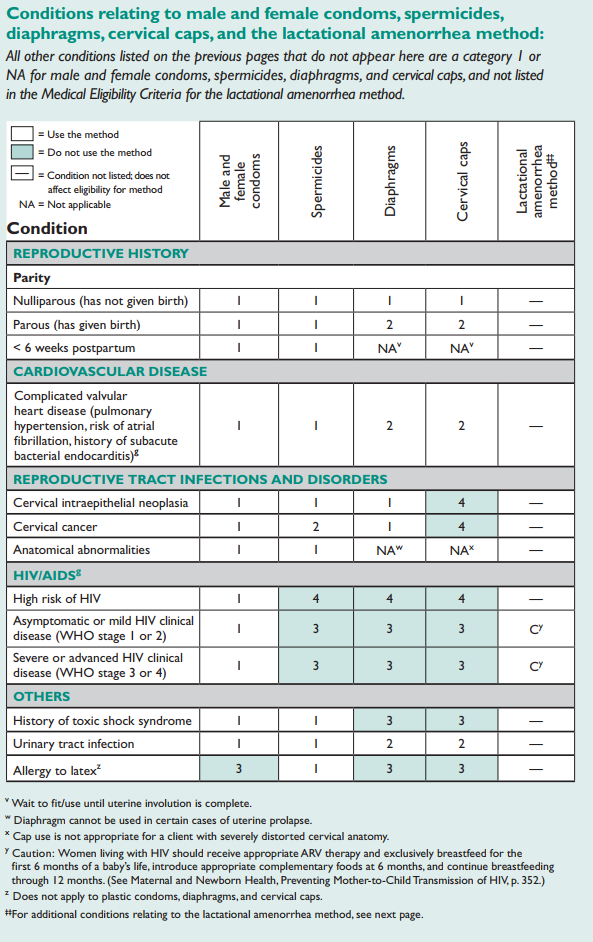
‡‡Additional conditions relating to lactational amenorrhea method:
Conditions affecting the newborn that may make breastfeeding difficult: Congenital deformities of the mouth, jaw, or palate; newborns who are small-for-date or premature and needing intensive neonatal care; and certain metabolic disorders.
Medication used during breastfeeding: To protect infant health, breastfeeding is not recommended for women using such drugs as anti-metabolites, bromocriptine, certain anticoagulants, corticosteroids (high doses), cyclosporine, ergotamine, lithium, mood-altering drugs, radioactive drugs, and reserpine.